G2C::Genetics
GRIT knock-out mouse
S.G.N. Grant and the G2C Consortium
Corresponding email: Seth.Grant@ed.ac.uk
G2CMine Data Warehouse
| Arhgap32 @ G2CMine |
Genetic and Genomic Information
| Gene symbol | Arhgap32 |
| MGI ID | MGI:2450166 |
| G2Cdb mouse | G00000017 |
| Ensembl mouse | ENSMUSG00000041444 |
| G2Cdb human | G00000042 |
| Ensembl human | ENSG00000134909 |
G2CMine Data Warehouse
G2CMine integrates the scientific findings of the Genes to Cognition Programme that utilised neuroproteomics, psychiatric genetics, high-throughput mouse gene targeting combined with behavioural and electrophysiological phenotyping and informatics in order to develop a general strategy for understanding cognition at the molecular, cellular and systems neuroscience levels.
G2CMine provides comprehensive Gene Ontology, Mammalian Phenotype Ontology, Human Phenotype Ontology, UniProt, genetic and protein interactions, and regional mouse brain expression results, together with the phenotyping results of the G2C Programme.
Mutation
A mouse embryonic stem (ES) cell line (XG279, strain 129/Ola) with an insertional mutation in Arhgap32 was obtained from BayGenomics (baygenomics.ucsf.edu/). The insertional mutation in XG279 by the gene-trapping vector, pGT1lxf, that was designed to create an in-frame fusion between the 5' exons of the trapped gene and a reporter, β-geo (a fusion of β-galactosidase and neomycin phosphotransferase II) occurred in intron 19-20. Thus, the gene-trapped locus is predicted to yield a fusion transcript containing exons 1–19 of Arhgap32 and β-geo.
The ES cells were injected into C57/BL6 blastocysts to create chimeric mice, which were bred with 129S5 mice to generate heterozygous (+/–) Arhgap32-mutant mice. Those F1 heterozygous mice had been backcrossed with 129S5 mice for 1-2 times before being used for intercrossing to produce homozygous mutants.
Location of Arhgap32 gene trap. Arhgap32 is a 22 exon protein and contains Phox, SH3 and RhoGAP domains (top). The Arhgap32 gene trap is located in intron 19-20.
Genotyping
Genomic DNA was isolated from ES cells or mouse tissues by Wizard SV 96 Genomic DNA purification system (Promega Cat A2371). Genotyping PCR consisted of a 1.7kb product amplified from the wild-type (wt) allele using a forward primer A (5'- GTCCCTTACTTCTCTCCATG -3') upstream of the cassette and a reverse primer B (5'- CTGAGCTACAGTATTGGAGAC -3') downstream of the trap. A 760bp product was amplified from the targeted allele using primer A with reverse primer C (5’- CATCCACTACTCAGTGCAGTG -3’), within the β-geo cassette. After enzymatic amplification for 30 cycles (45 seconds at 94 °C, 1 minute at 59 °C, and 2 minutes at 72 °C), the PCR products were size-fractionated on a 1% agarose gel in 1x Tris borate-EDTA buffer.
Primers used for genotyping (a,b & c). PCR genotyping of gene trap Arhgap32 mice using a common forward primer, a, and reverse primers b and c to amplify the wt and mutant alleles respectively.
Expression
Total RNA (100µg) was isolated from ES cells with RNeasy Mini kit (Qiagen, Cat 74104). RT-PCR was performed by generating first strand cDNA using Oligo(dT)12-18 primer (Invitrogen, Cat 18418-012) and superscript II reverse transcriptase (Invitrogen, Cat 18064-071). RT-PCR to detect a loss of ArhGAP32 RNA consisted of a 238bp product amplified from the wild-type (wt) cDNA using a forward primer Z (5’- CTGAAGAAGTAGAGTTGCCAG -3’) which anneals to exon 20 and a reverse primer Y (5’- CTCTTCAGTGTCTGACTTGAG -3’) which anneals to exon 21. To locate the gene trap a 430bp product was amplified from the gene trap cDNA using forward primer X (5’-GTCAGTTCTCCTATTGTGACG-3’) with reverse Primer W (5’- GATCCTCTAGAGTCCAGATCTG -3’), within the β-geo cassette. After enzymatic amplification for 35 cycles (45 seconds at 94 °C, 1 minute at 55 °C, and 1 minute at 72 °C), the PCR products were size-fractionated on a 2% agarose gel in 1x Tris borate-EDTA buffer.
Primers used for RT-PCR (w,x,y&z) RT-PCR - forward primer x (in exon 17) and reverse primer w (within the gene trap) are used to confirm the location of the trap (upper image). RT-PCR - forward primer, z (in exon 20) and reverse primer, y (in exon 21) are used to detect loss of RNA. Amplification is weaker in homozygous mice where the gene trap cassette has been inserted (lower image)
Breeding
Birth of Grit-/- mice followed Mendelian ratios with 27% of offspring being homozygous knockouts. Genotypes of 3-week-old pups from Grit+/- intercrosses identified 69 wt, 93 Grit+/- and 61 Grit-/- progeny (Χ2 p= 0.035). Male and female Grit-/- mice developed normally to adulthood, exhibited normal body size and no gross abnormalities. Grit mice were maintained by backcrossing onto the C57BL/6J background; heterozygous males and females were fertile and used to set up intercrosses to generate homozygous and wildtype mice to study.
Overview
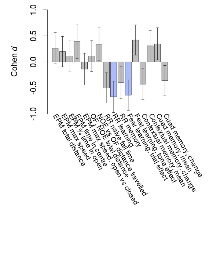
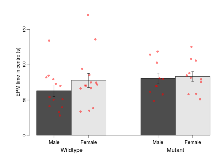
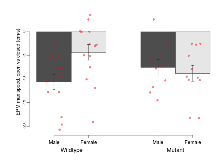
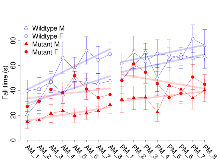
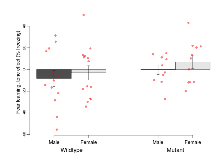
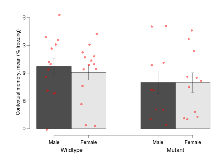
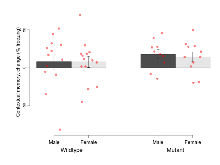
Mutant mice showed little overall behavioural difference from wildtypes, with two of 16 behaviour variables significantly impacted in these mutant mice. In the rotarod task, one behavioural variable, RR learning, was significantly decreased in mutants. In the fear learning task, one behavioural variable, fear learning trial effect, was significantly decreased in mutants. Elevated plus maze, open field/novel object exploration, contextual memory and cued memory tasks were unaffected. Definitions of variables may be found below.
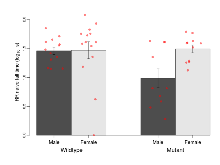
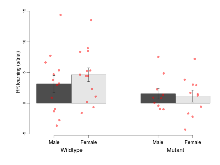
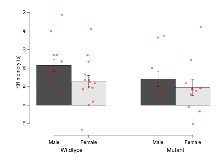
The G2CMine data warehouse provides cohort summaries and individual mouse observations from the GRIT knock-out line phenotyping.Variables shown are: EPM total distance, Total distance (cm) travelled in any arm or central zone of the EPM. EPM max speed, Maximum speed (cm/s) travelled in any arm or central zone of the EPM. EPM % time in open, Percentage of time in the open or closed arms of the EPM spent in open arms. EPM time in centre, Total time (s) spent in the central zone of the EPM. EPM max speed, open vs closed, Difference between the maximum speed (cm/s) observed in the open arms and the closed arms of the EPM. OF, NOE total distance, Total distance travelled (log₁₀ cm) during initial exposure to the open field and in presence of the novel object. NOE vs OF distance travelled, Difference in distance travelled (cm) in presence of the novel object and during initial exposure to open field. RR naive fall time, Fall time on accelerating rotarod (log₁₀ s), naive performance in session 1. RR learning, Learning on rotarod, measured as increase in fall time per trial (s/trial) in session 1. RR memory, Memory on rotarod, measured as excess fall time at middle of session 2 relative to middle of session 1. Fear learning, trial effect, Fear learning, measured as extra % time freezing before third trial compared to % time freezing before first trial. Fear learning, tone effect, Fear learning, measured as increase in % time freezing due to third tone compared to increase in % time freezing due to first tone. Contextual memory, mean, Contextual memory, measured as difference in % time freezing during first 120 s re-exposure to the box compared to first 120 s in the box on previous day. Contextual memory, change, Contextual memory, measured as increase in % time spent freezing from first time bin of 30 s to fourth bin of 30 s during 120 s re-exposure to the box. Cued memory, mean, Cued memory, measured as increase in % time spent freezing during 120 s of tone re-exposure compared to increase in % time spent freezing during initial tone on previous day. Cued memory, change, Cued memory, measured as increase in % time spent freezing from first time bin of 30 s to fourth bin of 30 s during 120 s re-exposure to the tone.
| Variable | Units | Wildtype M (n=13) | Wildtype F (n=14) | Mutant M (n=10) | Mutant F (n=11) | P(sex×mutation) | P(mutation) |
|---|---|---|---|---|---|---|---|
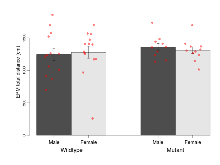
| EPM total distance | cm | 1244 (95) | 1279 (93) | 1360 (56) | 1311 (51) | 0.62 | 0.39 |
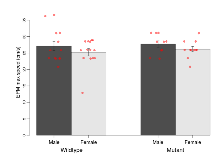
| EPM max speed | cm/s | 27.2 (1.4) | 25.3 (1.1) | 27.8 (1) | 26.2 (0.8) | 0.89 | 0.51 |
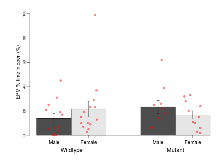
| EPM % time in open | % | 13.9 (3.8) | 21.6 (6.5) | 23.3 (5.3) | 16.7 (3.4) | 0.17 | 0.71 |
| EPM time in centre | s | 63.1 (7.9) | 77.8 (10) | 80.5 (7.6) | 83.3 (6.9) | 0.5 | 0.2 |
| EPM max speed, open vs closed | cm/s | -10.6 (1.6) | -4.4 (1.6) | -7.6 (1.6) | -8.8 (1.7) | 0.03 * | 0.61 |
| OF, NOE total distance | log10 cm | 3.61 (0.04) | 3.69 (0.04) | 3.6 (0.04) | 3.72 (0.03) | 0.56 | 0.7 |
| NOE vs OF distance travelled | cm | -1070 (180) | -1100 (200) | -610 (220) | -1070 (210) | 0.3 | 0.26 |
| RR naive fall time | log10 s | 1.46 (0.06) | 1.47 (0.15) | 0.99 (0.16) | 1.49 (0.07) | 0.044 * | 0.079 |
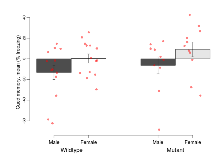
| RR learning | s/trial | 3.1 (1.4) | 4.6 (1.1) | 1.5 (0.9) | 1.2 (1) | 0.42 | 0.03 * |
| RR memory | s | 21.5 (3.3) | 12.6 (3.4) | 14.2 (4.2) | 9.5 (4.2) | 0.58 | 0.18 |
| Fear learning, trial effect | % freezing | 44.2 (5.5) | 41.3 (6.3) | 27.5 (6.9) | 31.2 (6.2) | 0.6 | 0.041 * |
| Fear learning, tone effect | % freezing | -8.2 (7.3) | -3.1 (6.5) | -0.3 (4.3) | 6.8 (5.9) | 0.88 | 0.17 |
| Contextual memory, mean | % freezing | 44.8 (6.1) | 40.5 (5.4) | 33.3 (8.2) | 33.2 (7) | 0.76 | 0.16 |
| Contextual memory, change | % freezing | 8.2 (9.9) | 7.7 (7.4) | 17.9 (6) | 14.4 (6.4) | 0.85 | 0.32 |
| Cued memory, mean | % freezing | -13.2 (6.9) | 0.3 (4.4) | -6.4 (8.2) | 9.1 (7.1) | 0.88 | 0.24 |
| Cued memory, change | % freezing | 6.7 (7.2) | -3 (8) | -9.7 (7.1) | -5.7 (7.7) | 0.38 | 0.24 |
Elevated Plus Maze
Variables shown are: EPM total distance, Total distance (cm) travelled in any arm or central zone of the EPM. EPM max speed, Maximum speed (cm/s) travelled in any arm or central zone of the EPM. EPM % time in open, Percentage of time in the open or closed arms of the EPM spent in open arms. EPM time in centre, Total time (s) spent in the central zone of the EPM. EPM max speed, open vs closed, Difference between the maximum speed (cm/s) observed in the open arms and the closed arms of the EPM.
| Variable | Units | Wildtype M (n=13) | Wildtype F (n=14) | Mutant M (n=10) | Mutant F (n=11) | P(sex×mutation) | P(mutation) |
|---|---|---|---|---|---|---|---|
| EPM total distance | cm | 1244 (95) | 1279 (93) | 1360 (56) | 1311 (51) | 0.62 | 0.39 |
| EPM max speed | cm/s | 27.2 (1.4) | 25.3 (1.1) | 27.8 (1) | 26.2 (0.8) | 0.89 | 0.51 |
| EPM % time in open | % | 13.9 (3.8) | 21.6 (6.5) | 23.3 (5.3) | 16.7 (3.4) | 0.17 | 0.71 |
| EPM time in centre | s | 63.1 (7.9) | 77.8 (10) | 80.5 (7.6) | 83.3 (6.9) | 0.5 | 0.2 |
| EPM max speed, open vs closed | cm/s | -10.6 (1.6) | -4.4 (1.6) | -7.6 (1.6) | -8.8 (1.7) | 0.03 * | 0.61 |
Open Field/Novel Object
Variables shown are: OF, NOE total distance, Total distance travelled (log₁₀ cm) during initial exposure to the open field and in presence of the novel object. NOE vs OF distance travelled, Difference in distance travelled (cm) in presence of the novel object and during initial exposure to open field.
| Variable | Units | Wildtype M (n=13) | Wildtype F (n=14) | Mutant M (n=10) | Mutant F (n=11) | P(sex×mutation) | P(mutation) |
|---|---|---|---|---|---|---|---|
| OF, NOE total distance | log10 cm | 3.61 (0.04) | 3.69 (0.04) | 3.6 (0.04) | 3.72 (0.03) | 0.56 | 0.7 |
| NOE vs OF distance travelled | cm | -1070 (180) | -1100 (200) | -610 (220) | -1070 (210) | 0.3 | 0.26 |
Rotarod
Variables shown are: RR naive fall time, Fall time on accelerating rotarod (log₁₀ s), naive performance in session 1. RR learning, Learning on rotarod, measured as increase in fall time per trial (s/trial) in session 1. RR memory, Memory on rotarod, measured as excess fall time at middle of session 2 relative to middle of session 1.
| Variable | Units | Wildtype M (n=13) | Wildtype F (n=14) | Mutant M (n=10) | Mutant F (n=11) | P(sex×mutation) | P(mutation) |
|---|---|---|---|---|---|---|---|
| RR naive fall time | log10 s | 1.46 (0.06) | 1.47 (0.15) | 0.99 (0.16) | 1.49 (0.07) | 0.044 * | 0.079 |
| RR learning | s/trial | 3.1 (1.4) | 4.6 (1.1) | 1.5 (0.9) | 1.2 (1) | 0.42 | 0.03 * |
| RR memory | s | 21.5 (3.3) | 12.6 (3.4) | 14.2 (4.2) | 9.5 (4.2) | 0.58 | 0.18 |
Fear Conditioning
Variables shown are: Fear learning, trial effect, Fear learning, measured as extra % time freezing before third trial compared to % time freezing before first trial. Fear learning, tone effect, Fear learning, measured as increase in % time freezing due to third tone compared to increase in % time freezing due to first tone. Contextual memory, mean, Contextual memory, measured as difference in % time freezing during first 120 s re-exposure to the box compared to first 120 s in the box on previous day. Contextual memory, change, Contextual memory, measured as increase in % time spent freezing from first time bin of 30 s to fourth bin of 30 s during 120 s re-exposure to the box. Cued memory, mean, Cued memory, measured as increase in % time spent freezing during 120 s of tone re-exposure compared to increase in % time spent freezing during initial tone on previous day. Cued memory, change, Cued memory, measured as increase in % time spent freezing from first time bin of 30 s to fourth bin of 30 s during 120 s re-exposure to the tone.
| Variable | Units | Wildtype M (n=13) | Wildtype F (n=14) | Mutant M (n=10) | Mutant F (n=11) | P(sex×mutation) | P(mutation) |
|---|---|---|---|---|---|---|---|
| Fear learning, trial effect | % freezing | 44.2 (5.5) | 41.3 (6.3) | 27.5 (6.9) | 31.2 (6.2) | 0.6 | 0.041 * |
| Fear learning, tone effect | % freezing | -8.2 (7.3) | -3.1 (6.5) | -0.3 (4.3) | 6.8 (5.9) | 0.88 | 0.17 |
| Contextual memory, mean | % freezing | 44.8 (6.1) | 40.5 (5.4) | 33.3 (8.2) | 33.2 (7) | 0.76 | 0.16 |
| Contextual memory, change | % freezing | 8.2 (9.9) | 7.7 (7.4) | 17.9 (6) | 14.4 (6.4) | 0.85 | 0.32 |
| Cued memory, mean | % freezing | -13.2 (6.9) | 0.3 (4.4) | -6.4 (8.2) | 9.1 (7.1) | 0.88 | 0.24 |
| Cued memory, change | % freezing | 6.7 (7.2) | -3 (8) | -9.7 (7.1) | -5.7 (7.7) | 0.38 | 0.24 |
- Overview
- Basal Synaptic Transmission
- Paired Pulse Facilitation
- Theta Burst Stimulation
- Long Term Potentiation
Overview
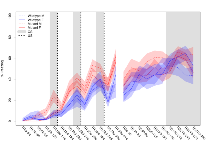
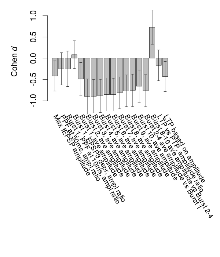
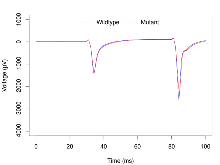
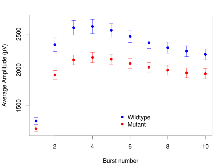
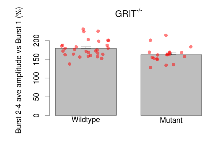
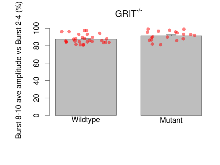
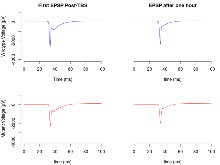
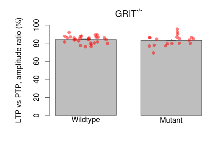
Mutant slices showed medium overall electrophysiological difference from wildtype slices, with no electrophysiological variables significantly affected by this mutation. Note that in the theta burst analysis, the tenth burst is analysed as representative of individual burst amplitude.
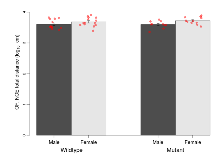
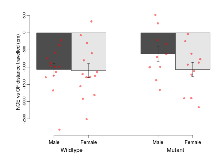
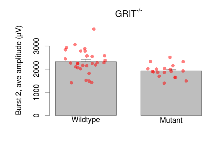
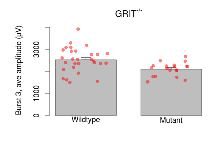
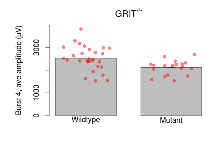
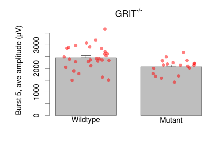
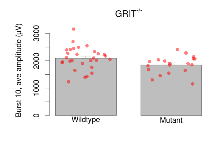
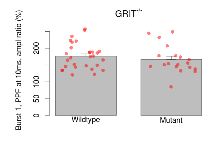
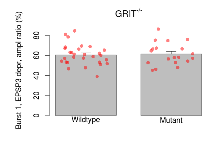
The G2CMine data warehouse provides slice group summaries and individual mouse observations from the GRIT knock-out line phenotyping.Variables shown are: Max fEPSP amplitude, Maximum field excitatory postsynaptic potential (fEPSP) amplitude. PPF at 50ms, ampl ratio, Paired pulse facilitation (PPF), pulses separated by 50ms, amplitude ratio. Burst 1, PPF at 10ms, ampl ratio, Paired pulse facilitation (PPF), pulses separated by 10ms, amplitude ratio, observed during first two pulses of the first 100Hz burst during theta-burst stimulation. Burst 1, EPSP3 depr, ampl ratio, Depression observed in third fEPSP relative to the second fEPSP of the first 100Hz burst, amplitude ratio. Burst 1, ave amplitude, Average amplitude of four fEPSPs in first burst. Burst 2, ave amplitude, Average amplitude of four fEPSPs in second burst. Burst 3, ave amplitude, Average amplitude of four fEPSPs in third burst. Burst 4, ave amplitude, Average amplitude of four fEPSPs in fourth burst. Burst 5, ave amplitude, Average amplitude of four fEPSPs in fifth burst. Burst 6, ave amplitude, Average amplitude of four fEPSPs in sixth burst. Burst 7, ave amplitude, Average amplitude of four fEPSPs in seventh burst. Burst 8, ave amplitude, Average amplitude of four fEPSPs in eighth burst. Burst 9, ave amplitude, Average amplitude of four fEPSPs in ninth burst. Burst 10, ave amplitude, Average amplitude of four fEPSPs in tenth burst. Burst 2-4 ave amplitude vs Burst 1, Facilitation observed in average amplitude of bursts 2-4, relative to average amplitude of burst 1. Burst 8-10 ave amplitude vs Burst 2-4, Depression observed in average amplitude of bursts 8-10, relative to average amplitude of bursts 2-4. LTP vs PTP, amplitude ratio, Reduction in potentiation from immediately after theta-burst stimulation to one hour later, fEPSP amplitude ratio. LTP based on amplitude, Long term potentiation, ratio of amplitudes of fEPSPs in test pathway and control pathway.
| Variable | Units | Wildtype slices (animals) | Wildtype mean (SEM) | Mutant slices (animals) | Mutant mean (SEM) | P(animals) | P(mutation) |
|---|---|---|---|---|---|---|---|
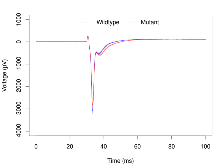
| Max fEPSP amplitude | µV | 29 (8) | 3267 (121) | 19 (5) | 3037 (80) | 0.00048 *** | 0.39 |
| PPF at 50ms, ampl ratio | % | 29 (8) | 172.7 (3.4) | 19 (5) | 167.8 (5) | 0.41 | 0.43 |
| Burst 1, PPF at 10ms, ampl ratio | % | 29 (8) | 176.3 (7.1) | 19 (5) | 166.4 (9.6) | 0.29 | 0.46 |
| Burst 1, EPSP3 depr, ampl ratio | % | 29 (8) | 60.6 (1.9) | 19 (5) | 61.5 (2.6) | 0.079 | 0.83 |
| Burst 1, ave amplitude | µV | 29 (8) | 1377 (49) | 19 (5) | 1267 (35) | 0.0038 ** | 0.29 |
| Burst 2, ave amplitude | µV | 29 (8) | 2326 (97) | 19 (5) | 1925 (65) | 0.0018 ** | 0.069 |
| Burst 3, ave amplitude | µV | 29 (8) | 2525 (103) | 19 (5) | 2100 (70) | 0.0021 ** | 0.07 |
| Burst 4, ave amplitude | µV | 29 (8) | 2524 (99) | 19 (5) | 2116 (73) | 0.0024 ** | 0.071 |
| Burst 5, ave amplitude | µV | 29 (8) | 2451 (94) | 19 (5) | 2071 (74) | 0.0015 ** | 0.084 |
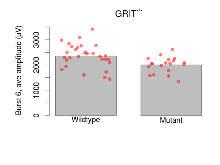
| Burst 6, ave amplitude | µV | 29 (8) | 2359 (87) | 19 (5) | 2004 (72) | 0.0012 ** | 0.086 |
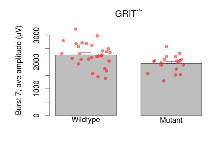
| Burst 7, ave amplitude | µV | 29 (8) | 2269 (81) | 19 (5) | 1949 (71) | 0.0017 ** | 0.096 |
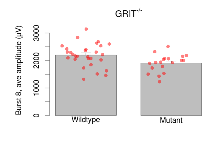
| Burst 8, ave amplitude | µV | 29 (8) | 2198 (77) | 19 (5) | 1907 (72) | 0.0024 ** | 0.11 |
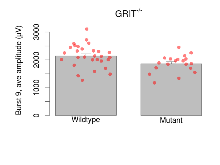
| Burst 9, ave amplitude | µV | 29 (8) | 2142 (76) | 19 (5) | 1860 (72) | 0.0038 ** | 0.11 |
| Burst 10, ave amplitude | µV | 29 (8) | 2094 (76) | 19 (5) | 1844 (74) | 0.0046 ** | 0.16 |
| Burst 2-4 ave amplitude vs Burst 1 | % | 29 (8) | 179.3 (4.2) | 19 (5) | 162.5 (4.8) | 0.14 | 0.054 |
| Burst 8-10 ave amplitude vs Burst 2-4 | % | 29 (8) | 87.6 (0.9) | 19 (5) | 91.4 (1.2) | 0.21 | 0.054 |
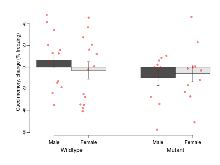
| LTP vs PTP, amplitude ratio | % | 29 (8) | 84.1 (0.7) | 19 (5) | 83.3 (1.4) | 0.067 | 0.66 |
| LTP based on amplitude | % | 29 (8) | 179.5 (4.7) | 19 (5) | 169.5 (4.4) | 0.026 * | 0.3 |
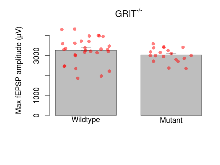
Basal Synaptic Transmission
Variables shown are: Max fEPSP amplitude, Maximum field excitatory postsynaptic potential (fEPSP) amplitude.
| Variable | Units | Wildtype slices (animals) | Wildtype mean (SEM) | Mutant slices (animals) | Mutant mean (SEM) | P(animals) | P(mutation) |
|---|---|---|---|---|---|---|---|
| Max fEPSP amplitude | µV | 29 (8) | 3267 (121) | 19 (5) | 3037 (80) | 0.00048 *** | 0.39 |
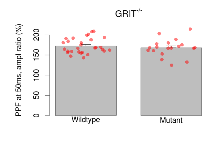
Paired Pulse Facilitation
Variables shown are: PPF at 50ms, ampl ratio, Paired pulse facilitation (PPF), pulses separated by 50ms, amplitude ratio.
| Variable | Units | Wildtype slices (animals) | Wildtype mean (SEM) | Mutant slices (animals) | Mutant mean (SEM) | P(animals) | P(mutation) |
|---|---|---|---|---|---|---|---|
| PPF at 50ms, ampl ratio | % | 29 (8) | 172.7 (3.4) | 19 (5) | 167.8 (5) | 0.41 | 0.43 |
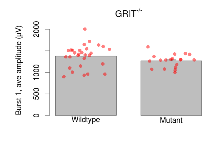
Theta Burst Stimulation
Variables shown are: Burst 1, PPF at 10ms, ampl ratio, Paired pulse facilitation (PPF), pulses separated by 10ms, amplitude ratio, observed during first two pulses of the first 100Hz burst during theta-burst stimulation. Burst 1, EPSP3 depr, ampl ratio, Depression observed in third fEPSP relative to the second fEPSP of the first 100Hz burst, amplitude ratio. Burst 1, ave amplitude, Average amplitude of four fEPSPs in first burst. Burst 2, ave amplitude, Average amplitude of four fEPSPs in second burst. Burst 3, ave amplitude, Average amplitude of four fEPSPs in third burst. Burst 4, ave amplitude, Average amplitude of four fEPSPs in fourth burst. Burst 5, ave amplitude, Average amplitude of four fEPSPs in fifth burst. Burst 6, ave amplitude, Average amplitude of four fEPSPs in sixth burst. Burst 7, ave amplitude, Average amplitude of four fEPSPs in seventh burst. Burst 8, ave amplitude, Average amplitude of four fEPSPs in eighth burst. Burst 9, ave amplitude, Average amplitude of four fEPSPs in ninth burst. Burst 10, ave amplitude, Average amplitude of four fEPSPs in tenth burst. Burst 2-4 ave amplitude vs Burst 1, Facilitation observed in average amplitude of bursts 2-4, relative to average amplitude of burst 1. Burst 8-10 ave amplitude vs Burst 2-4, Depression observed in average amplitude of bursts 8-10, relative to average amplitude of bursts 2-4.
| Variable | Units | Wildtype slices (animals) | Wildtype mean (SEM) | Mutant slices (animals) | Mutant mean (SEM) | P(animals) | P(mutation) |
|---|---|---|---|---|---|---|---|
| Burst 1, PPF at 10ms, ampl ratio | % | 29 (8) | 176.3 (7.1) | 19 (5) | 166.4 (9.6) | 0.29 | 0.46 |
| Burst 1, EPSP3 depr, ampl ratio | % | 29 (8) | 60.6 (1.9) | 19 (5) | 61.5 (2.6) | 0.079 | 0.83 |
| Burst 1, ave amplitude | µV | 29 (8) | 1377 (49) | 19 (5) | 1267 (35) | 0.0038 ** | 0.29 |
| Burst 2, ave amplitude | µV | 29 (8) | 2326 (97) | 19 (5) | 1925 (65) | 0.0018 ** | 0.069 |
| Burst 3, ave amplitude | µV | 29 (8) | 2525 (103) | 19 (5) | 2100 (70) | 0.0021 ** | 0.07 |
| Burst 4, ave amplitude | µV | 29 (8) | 2524 (99) | 19 (5) | 2116 (73) | 0.0024 ** | 0.071 |
| Burst 5, ave amplitude | µV | 29 (8) | 2451 (94) | 19 (5) | 2071 (74) | 0.0015 ** | 0.084 |
| Burst 6, ave amplitude | µV | 29 (8) | 2359 (87) | 19 (5) | 2004 (72) | 0.0012 ** | 0.086 |
| Burst 7, ave amplitude | µV | 29 (8) | 2269 (81) | 19 (5) | 1949 (71) | 0.0017 ** | 0.096 |
| Burst 8, ave amplitude | µV | 29 (8) | 2198 (77) | 19 (5) | 1907 (72) | 0.0024 ** | 0.11 |
| Burst 9, ave amplitude | µV | 29 (8) | 2142 (76) | 19 (5) | 1860 (72) | 0.0038 ** | 0.11 |
| Burst 10, ave amplitude | µV | 29 (8) | 2094 (76) | 19 (5) | 1844 (74) | 0.0046 ** | 0.16 |
| Burst 2-4 ave amplitude vs Burst 1 | % | 29 (8) | 179.3 (4.2) | 19 (5) | 162.5 (4.8) | 0.14 | 0.054 |
| Burst 8-10 ave amplitude vs Burst 2-4 | % | 29 (8) | 87.6 (0.9) | 19 (5) | 91.4 (1.2) | 0.21 | 0.054 |
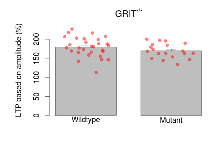
Long Term Potentiation
Variables shown are: LTP vs PTP, amplitude ratio, Reduction in potentiation from immediately after theta-burst stimulation to one hour later, fEPSP amplitude ratio. LTP based on amplitude, Long term potentiation, ratio of amplitudes of fEPSPs in test pathway and control pathway.
| Variable | Units | Wildtype slices (animals) | Wildtype mean (SEM) | Mutant slices (animals) | Mutant mean (SEM) | P(animals) | P(mutation) |
|---|---|---|---|---|---|---|---|
| LTP vs PTP, amplitude ratio | % | 29 (8) | 84.1 (0.7) | 19 (5) | 83.3 (1.4) | 0.067 | 0.66 |
| LTP based on amplitude | % | 29 (8) | 179.5 (4.7) | 19 (5) | 169.5 (4.4) | 0.026 * | 0.3 |
Behaviour raw data
| Animals | View |
| Elevated Plus Maze | View |
| Open Field | View |
| Novel Object Exploration | View |
| Rotarod | View |
Hippocampal slice electrophysiology raw data
| Animals and Brain slices | View |
| Field EPSP (Maximum) | View |
| Paired Pulse Facilitation | View |
| Long Term Potentiation | View |
| Data Files | View |