G2C::Neuroproteomics Background
Proteomics is a young and rapidly evolving discipline of biochemistry but it has a well defined objective: the study of the proteome, the complete set of proteins produced by a certain organism. The technologies by which this is achieved can vary a lot but all share the ability of managing large-scale protein separation and identification. Since proteins are much more dynamic than nucleic acids proteomics goes beyond the mere identification of proteins, it also has to deal with protein modification, expression regulation and how proteins interact with each other.
In our lab we are especially interested in a subset of a species proteome, the one involved in synapse functioning. We believe that an accurate explanation of the molecular mechanisms underlying synapse functioning will suppose a huge step forward in the understanding of the brain and it's cognitive abilities.
In our lab we are especially interested in a subset of a species proteome, the one involved in synapse functioning. We believe that an accurate explanation of the molecular mechanisms underlying synapse functioning will suppose a huge step forward in the understanding of the brain and it's cognitive abilities.
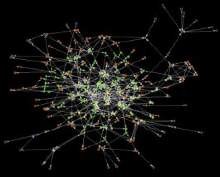
Through this analysis a picture emerged of 75 or more proteins that could be broadly categorised into five classes: neurotransmitter receptors, cell adhesion molecules, adaptors, signaling enzymes and cytoskeletal proteins (10862698). A major surprise in this study was that at least 27 proteins from the complexes were known to be required for synaptic plasticity and 18 for learning in rodents, and were from each of the five classes of complex components. Thus the organisation of these proteins into these multiprotein complexes suggests that they work together in a large 'machine', not unlike many other multiprotein molecular machines.
The importance of this concept is that it removes the focus of interest from individual molecules and places it on the function of the overall machine. We propose that these complexes are a 'device' for detecting patterns of synaptic activity and for converting this information into intracellular signals that store the information in the cell. In this way, electrical information can be translated into cellular memory.
These properties were at the basis of Donald Hebb's postulated mechanism of memory, and these complexes have been described as Hebbosomes; multiprotein complexes that convert patterns of neuronal activity into cellular changes underlying learning.
Nowadays those proteins present in the synapse are much better defined. Our work contributed to the understanding of the components at the post-synapse (16635246; 14532281; 16507876), which is better characterised than the pre-synaptic site, although some papers describe this structure (16269012; 15378707). Therefore, the future goals in synapse proteomics will have to deal with more functional issues. Phosphoproteomics is the most common functional analysis of a certain proteome and it has also been performed in the study of the synapse (18056256; 16635246; 17114649). These data has increased the existing knowledge of synapse protein phosphorylation and has enforced the idea that the highly interconnected signalling network present in the synapse relays very much in phosphorylation events for its proper functioning.